Multiplex vs Single Rapid Tests: Which Is More Cost-Effective for Clinical and Procurement Needs?
Multiplex rapid tests are generally more cost-effective in high-throughput and multi-pathogen scenarios, while single tests remain economical for targeted, low-volume use. Choosing between multiplex vs single rapid test depends on diagnostic scope, workflow efficiency, and total cost per clinical decision.
What Are Multiplex and Single Rapid Tests?
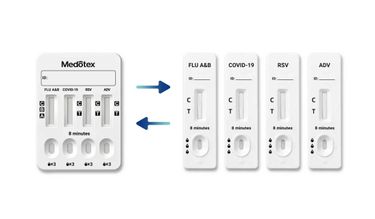
A multiplex rapid test detects multiple pathogens from a single sample, typically using lateral flow or similar immunoassay technologies. These tests are widely used in respiratory diagnostics where symptoms overlap.
A single rapid test detects one specific pathogen per test. It is often used when a particular infection is strongly suspected or confirmed.
Understanding this distinction is essential when evaluating multiplex rapid test vs single test strategies in clinical and procurement settings.
Key Differences Between Multiplex and Single Rapid Tests
| Comparison Factor | Multiplex Rapid Test | Single Rapid Test |
|---|---|---|
| Diagnostic Scope | Detects multiple pathogens in one test | Detects one specific pathogen per test |
| Testing Workflow | One-step testing, reduces repeat assays | May require sequential testing for different pathogens |
| Turnaround Time | Single run provides comprehensive results | Multiple tests may extend total diagnostic time |
| Resource Utilization | Lower overall use of consumables and staff time | Higher cumulative resource use if multiple tests are needed |
| Cost Efficiency | More cost-effective in multi-pathogen scenarios | Lower upfront cost per test but higher total cost in complex cases |
Cost Analysis: Which Is More Economical?
At the unit level, single tests are typically less expensive. However, total cost analysis often favors multiplex testing in multi-pathogen scenarios.
A cost-consequence analysis published in the Brazilian Journal of Infectious Diseases demonstrated that combined COVID-19 and influenza rapid testing significantly improved clinical and economic outcomes. In a modeled cohort of 1,000 patients, multiplex testing reduced missed diagnoses by 88–149 cases and over-diagnosis by 161–185 cases, while decreasing hospital bed utilization by 24%–34% and ICU occupancy by 16%–26%. Overall, the combined testing strategy also resulted in net cost savings at the population level.
These findings highlight the real-world economic advantages of multiplex rapid test vs single test strategies, particularly in high-volume clinical settings.
When to Choose Multiplex Rapid Tests
Multiplex testing is recommended when diagnostic efficiency and broad coverage are required.
Typical scenarios include:
- Respiratory infection season (e.g., influenza, COVID-19, RSV peaks)
- Outbreak investigations and high-risk populations
- Emergency and inpatient settings with undifferentiated symptoms
- High-throughput laboratories requiring workflow optimization
In these contexts, multiplex testing benefits include faster diagnosis, reduced repeat testing, and improved resource utilization. These advantages make multiplex vs single rapid test decisions lean toward multiplex solutions.
When to Choose Single Rapid Tests
Single tests remain appropriate in more targeted or constrained scenarios.
Common use cases include:
- Testing for a known or highly suspected pathogen
- Resource-limited or low-volume clinical settings
- Confirmatory or follow-up testing
- Large-scale screening programs focused on a single pathogen
In such cases, single tests may provide a cost-effective and straightforward solution within a multiplex rapid test vs single test framework.
Diagnostic Accuracy and Clinical Value
Both testing approaches can offer reliable performance when used appropriately. Diagnostic accuracy depends on factors such as specimen quality, timing, and assay design.
Multiplex tests are designed to maintain high specificity across multiple targets, though performance may vary between pathogens. Single tests may offer slightly optimized sensitivity for individual targets.
From a clinical standpoint, the choice between multiplex vs single rapid test should align with diagnostic goals rather than relying solely on perceived accuracy differences.
Regulatory and Compliance Considerations
Regulatory requirements apply equally to multiplex and single rapid tests. Products should meet standards such as ISO 13485, CE marking, or relevant national approvals.
For procurement teams, verifying compliance is essential regardless of whether a multiplex rapid test vs single test strategy is selected. Regulatory validation ensures consistent performance and reduces operational risk.
Future Trends: Why Multiplex Testing Is Growing
Multiplex testing is gaining adoption due to increasing demand for efficiency and comprehensive diagnostics. Advances in assay design and manufacturing have improved reliability and scalability.
As healthcare systems face higher testing volumes and more complex diagnostic needs, multiplex testing benefits become more evident. This trend suggests that multiplex vs single rapid test decisions will increasingly favor multiplex solutions, especially in respiratory and infectious disease management.
Conclusion
Choosing between multiplex vs single rapid test depends on clinical context, testing volume, and cost structure. Multiplex testing offers clear advantages in multi-pathogen and high-throughput scenarios, while single tests remain effective for targeted applications.
For healthcare providers and distributors seeking efficient diagnostic solutions, working with experienced manufacturers such as Medotex can support flexible testing strategies and scalable implementation.